Based On The Molecular Structures Shown In The Figure
Breaking News Today
Apr 03, 2025 · 6 min read
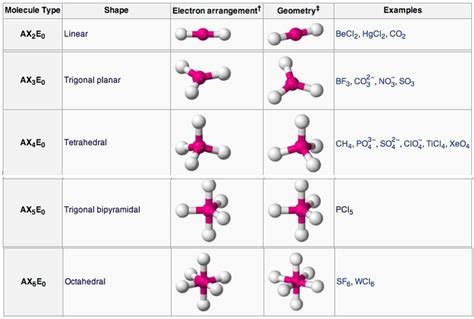
Table of Contents
Unveiling Molecular Mysteries: A Deep Dive into Molecular Structures
Analyzing molecular structures is fundamental to understanding chemistry, biology, and materials science. A single image, depicting various molecular structures, can unlock a wealth of information regarding chemical properties, reactivity, and potential applications. This article delves into the fascinating world of molecular structures, exploring how their unique arrangements dictate their behavior and functionality. While I cannot directly access or interpret a "figure" provided outside this context, I will provide a comprehensive framework for analyzing molecular structures, encompassing key concepts and examples.
Understanding the Basics: Key Concepts in Molecular Structure Analysis
Before embarking on a detailed analysis of specific structures, it’s crucial to establish a common understanding of fundamental concepts. These include:
1. Atomic Structure and Bonding:
The foundation of any molecular structure lies in the arrangement of atoms. Atoms, composed of protons, neutrons, and electrons, interact through chemical bonds. These bonds can be:
-
Covalent Bonds: Formed by the sharing of electrons between atoms. The strength and length of these bonds significantly influence molecular shape and reactivity. Examples include the strong bonds in methane (CH₄) or the slightly weaker bonds in ethene (C₂H₄).
-
Ionic Bonds: Result from the electrostatic attraction between oppositely charged ions. These bonds are generally stronger than covalent bonds in certain environments. Sodium chloride (NaCl), table salt, is a prime example.
-
Hydrogen Bonds: A special type of dipole-dipole interaction, these relatively weak bonds play a crucial role in biological systems, influencing the structure and function of proteins and DNA. Water's unique properties are largely attributed to its hydrogen bonding network.
-
Van der Waals Forces: Weak intermolecular forces that arise from temporary fluctuations in electron distribution. While individually weak, collectively they can significantly affect physical properties like boiling point and melting point.
2. Molecular Geometry and Shapes:
The three-dimensional arrangement of atoms in a molecule determines its geometry and shape. These shapes are crucial in dictating reactivity and properties. Common shapes include:
-
Linear: Atoms arranged in a straight line (e.g., CO₂).
-
Bent: Atoms arranged in a bent or V-shape (e.g., H₂O).
-
Trigonal Planar: Atoms arranged in a flat, triangular shape (e.g., BF₃).
-
Tetrahedral: Atoms arranged in a four-sided pyramid shape (e.g., CH₄).
-
Octahedral: Atoms arranged in an eight-sided shape (e.g., SF₆).
These shapes are often predicted using the Valence Shell Electron Pair Repulsion (VSEPR) theory, which posits that electron pairs repel each other and arrange themselves to minimize repulsion.
3. Isomerism: Same Formula, Different Structure
Isomers are molecules with the same molecular formula but different structural arrangements. Several types of isomerism exist:
-
Structural Isomers (Constitutional Isomers): These isomers differ in the connectivity of their atoms. For instance, butane (C₄H₁₀) exists as two structural isomers: n-butane and isobutane.
-
Stereoisomers: These isomers have the same connectivity but differ in the spatial arrangement of their atoms. Further categorized into:
-
Geometric Isomers (cis-trans isomers): Differ in the arrangement of substituents around a double bond or ring.
-
Enantiomers (optical isomers): Molecules that are mirror images of each other and non-superimposable. They exhibit optical activity, rotating plane-polarized light in opposite directions.
-
4. Functional Groups: The Reactive Centers
Functional groups are specific groups of atoms within a molecule that confer characteristic chemical properties. Identifying functional groups is vital in predicting reactivity and understanding biological activity. Common functional groups include:
-
Hydroxyl (-OH): Alcohols
-
Carbonyl (C=O): Aldehydes, ketones, carboxylic acids
-
Carboxyl (-COOH): Carboxylic acids
-
Amino (-NH₂): Amines
-
Ester (-COO-): Esters
Analyzing Molecular Structures: A Step-by-Step Approach
To effectively analyze a molecular structure (assuming it's presented visually), follow these steps:
-
Identify the Atoms: Begin by identifying each atom present in the molecule. Pay close attention to their symbols (e.g., C for carbon, O for oxygen, H for hydrogen).
-
Determine the Connectivity: Observe how the atoms are bonded together. Single, double, or triple bonds have distinct implications for the molecule's properties.
-
Assess the Molecular Geometry: Using VSEPR theory or other relevant models, predict the molecule's three-dimensional shape. Consider the number of electron pairs surrounding each atom.
-
Identify Functional Groups: Pinpoint any functional groups present. These groups often determine the molecule's reactivity and chemical behavior.
-
Analyze for Isomerism: Check for any isomers. If isomers exist, determine the type (structural, geometric, or enantiomeric). This often requires careful comparison of spatial arrangements.
-
Predict Properties: Based on the structure, predict some key physical and chemical properties. These might include boiling point, melting point, solubility, reactivity towards certain reagents, and potential biological activity.
Examples of Molecular Structure Analysis
Let's hypothetically analyze a few common molecular structures to illustrate the process:
Example 1: Methane (CH₄)
-
Atoms: One carbon atom, four hydrogen atoms.
-
Connectivity: Each hydrogen atom is singly bonded to the central carbon atom.
-
Geometry: Tetrahedral – the carbon atom is at the center of a tetrahedron, with the four hydrogen atoms located at the vertices.
-
Functional Groups: None. Methane is a simple hydrocarbon.
-
Isomers: No isomers exist for methane.
-
Properties: Methane is a gas at room temperature, nonpolar, and relatively unreactive.
Example 2: Ethanol (C₂H₅OH)
-
Atoms: Two carbon atoms, six hydrogen atoms, one oxygen atom.
-
Connectivity: One carbon atom is bonded to three hydrogen atoms and one carbon atom. The second carbon atom is bonded to two hydrogen atoms, one carbon atom, and one oxygen atom. The oxygen atom is bonded to a hydrogen atom.
-
Geometry: Tetrahedral around each carbon atom, bent around the oxygen atom.
-
Functional Groups: Hydroxyl group (-OH), making ethanol an alcohol.
-
Isomers: Structural isomers of ethanol exist, such as dimethyl ether.
-
Properties: Ethanol is a liquid at room temperature, polar, soluble in water, and has properties of both alcohols and hydrocarbons.
Example 3: Benzene (C₆H₆)
-
Atoms: Six carbon atoms, six hydrogen atoms.
-
Connectivity: The carbon atoms form a planar hexagonal ring with alternating single and double bonds (resonance structure). Each carbon atom is bonded to one hydrogen atom.
-
Geometry: Planar, hexagonal.
-
Functional Groups: Aromatic ring.
-
Isomers: Various substituted benzenes are possible, depending on the placement of substituents.
-
Properties: Benzene is a liquid at room temperature, nonpolar (although it has polar bonds), and is known for its aromatic properties and tendency to undergo electrophilic aromatic substitution reactions.
Conclusion: The Power of Molecular Structure Analysis
The ability to analyze molecular structures is paramount in various fields. Understanding the relationship between a molecule's structure and its properties allows for:
-
Drug Design: Design drugs targeting specific proteins or receptors.
-
Materials Science: Create new materials with desired properties.
-
Environmental Science: Assess the environmental impact of various chemicals.
-
Biological Research: Understand how biological molecules function.
By mastering the techniques of molecular structure analysis, scientists and researchers unlock a deeper understanding of the natural world and pave the way for advancements across many scientific disciplines. While this article provides a robust theoretical foundation, practical application requires experience and familiarity with specialized software and databases. Continuous learning and exploration are vital in this ever-evolving field.
Latest Posts
Latest Posts
-
Which Leader Supported Pan Arabism In The Mid Twentieth Century
Apr 04, 2025
-
Unit 7 Progress Check Mcq Ap Chem
Apr 04, 2025
-
Which Of The Following Is Not A Symptom Of Schizophrenia
Apr 04, 2025
-
Which Statement About Ethnocentrism Is Most Accurate
Apr 04, 2025
-
How Is The Energy Value Of Foods Determined
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Based On The Molecular Structures Shown In The Figure . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
