Fluid Electrolyte And Acid-base Balance Ati Quizlet
Breaking News Today
Mar 31, 2025 · 6 min read
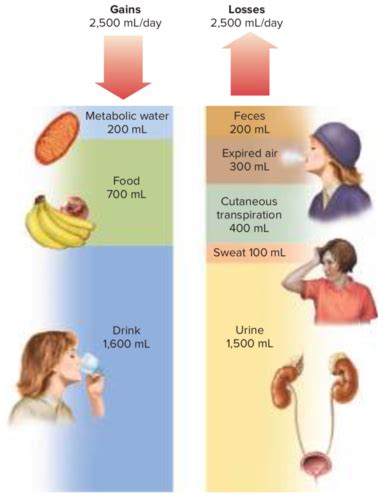
Table of Contents
- Fluid Electrolyte And Acid-base Balance Ati Quizlet
- Table of Contents
- Mastering Fluid, Electrolyte, and Acid-Base Balance: A Comprehensive Guide
- A. Fluid Intake and Output: A Delicate Balance
- B. Fluid Volume Imbalances: Recognizing the Signs and Symptoms
- A. Sodium (Na+): The Dominant Extracellular Cation
- B. Potassium (K+): The Dominant Intracellular Cation
- C. Calcium (Ca2+): Essential for Bones, Muscles, and Nerves
- D. Magnesium (Mg2+): A Multifaceted Mineral
- E. Chloride (Cl-): Maintaining Electrolyte Balance
- A. Acid-Base Buffers: The Body's First Line of Defense
- B. Respiratory Regulation of Acid-Base Balance
- C. Renal Regulation of Acid-Base Balance
- D. Acid-Base Imbalances: Recognizing the Patterns
- Latest Posts
- Latest Posts
- Related Post
Mastering Fluid, Electrolyte, and Acid-Base Balance: A Comprehensive Guide
Understanding fluid, electrolyte, and acid-base balance is crucial in healthcare. This in-depth guide explores these interconnected physiological processes, offering a comprehensive overview ideal for students and professionals alike. We'll delve into the intricacies of each component, examining their regulation, imbalances, and clinical implications. Think of this as your ultimate resource for conquering those challenging ATI quizzes and solidifying your knowledge.
I. Fluid Balance: The Foundation of Homeostasis
Fluid balance, the precise regulation of body fluid volume and composition, is paramount for overall health. Our bodies are primarily composed of water, distributed across various compartments: intracellular fluid (ICF), extracellular fluid (ECF) – further divided into interstitial fluid and intravascular fluid (plasma). Maintaining equilibrium between these compartments is essential for cellular function and organ system integrity.
A. Fluid Intake and Output: A Delicate Balance
Fluid intake primarily occurs through drinking and consuming foods, while output involves excretion through urine, feces, sweat, and insensible losses (respiration and skin evaporation). Hormonal regulation plays a critical role in maintaining this balance.
-
Antidiuretic Hormone (ADH): Produced by the hypothalamus and released by the posterior pituitary gland, ADH regulates water reabsorption in the kidneys. When blood volume is low or osmolality is high (concentrated), ADH release increases, leading to increased water reabsorption and concentrated urine.
-
Renin-Angiotensin-Aldosterone System (RAAS): Activated in response to decreased blood volume or pressure, this system stimulates aldosterone release from the adrenal cortex. Aldosterone promotes sodium and water reabsorption in the distal tubules and collecting ducts of the kidneys, increasing blood volume and pressure.
-
Atrial Natriuretic Peptide (ANP): Released by the atria in response to increased blood volume and pressure, ANP inhibits sodium and water reabsorption, promoting diuresis (increased urine production) and reducing blood volume.
B. Fluid Volume Imbalances: Recognizing the Signs and Symptoms
Disruptions in fluid intake and output, or malfunctions in the regulatory mechanisms, can lead to fluid volume imbalances.
-
Fluid Volume Deficit (Hypovolemia): Characterized by decreased extracellular fluid volume. Symptoms include thirst, decreased urine output, dry mucous membranes, hypotension, tachycardia, and weight loss.
-
Fluid Volume Excess (Hypervolemia): Characterized by increased extracellular fluid volume. Symptoms include edema, weight gain, distended neck veins, shortness of breath, hypertension, and crackles in the lungs.
II. Electrolyte Balance: Essential Ions for Cellular Function
Electrolytes are electrically charged minerals essential for numerous physiological processes, including nerve impulse transmission, muscle contraction, and fluid balance. Key electrolytes include sodium (Na+), potassium (K+), calcium (Ca2+), magnesium (Mg2+), and chloride (Cl-).
A. Sodium (Na+): The Dominant Extracellular Cation
Sodium plays a critical role in fluid balance, nerve impulse transmission, and muscle contraction. Sodium imbalance can significantly affect fluid volume and neurological function.
-
Hyponatremia (low sodium): Symptoms can range from mild nausea and headache to seizures and coma.
-
Hypernatremia (high sodium): Symptoms include thirst, altered mental status, seizures, and coma.
B. Potassium (K+): The Dominant Intracellular Cation
Potassium is essential for maintaining normal heart rhythm, muscle function, and nerve impulse transmission. Imbalances are particularly dangerous, especially for cardiac function.
-
Hypokalemia (low potassium): Symptoms include muscle weakness, fatigue, cardiac arrhythmias, and constipation.
-
Hyperkalemia (high potassium): Symptoms include muscle weakness, cardiac arrhythmias (potentially fatal), and nausea.
C. Calcium (Ca2+): Essential for Bones, Muscles, and Nerves
Calcium is crucial for bone health, muscle contraction, nerve impulse transmission, and blood clotting.
-
Hypocalcemia (low calcium): Symptoms include muscle spasms (tetany), seizures, and cardiac arrhythmias.
-
Hypercalcemia (high calcium): Symptoms include fatigue, constipation, kidney stones, and altered mental status.
D. Magnesium (Mg2+): A Multifaceted Mineral
Magnesium plays a vital role in numerous enzymatic reactions, muscle function, and nerve impulse transmission.
-
Hypomagnesemia (low magnesium): Symptoms include muscle weakness, tremors, and cardiac arrhythmias.
-
Hypermagnesemia (high magnesium): Symptoms include muscle weakness, hypotension, and respiratory depression.
E. Chloride (Cl-): Maintaining Electrolyte Balance
Chloride works in conjunction with other electrolytes, primarily sodium, to maintain fluid balance and acid-base homeostasis. Imbalances often occur in conjunction with sodium imbalances.
III. Acid-Base Balance: Maintaining pH Homeostasis
The body maintains a narrow range of blood pH (7.35-7.45) through complex buffering systems and regulatory mechanisms. Acid-base imbalances can significantly impact cellular function and organ system integrity.
A. Acid-Base Buffers: The Body's First Line of Defense
Buffers are substances that resist changes in pH by binding to or releasing hydrogen ions (H+). Important buffers include bicarbonate (HCO3-), phosphate, and proteins.
B. Respiratory Regulation of Acid-Base Balance
The lungs play a crucial role in regulating blood pH by controlling carbon dioxide (CO2) levels. CO2 reacts with water to form carbonic acid (H2CO3), which dissociates into H+ and HCO3-. Increased ventilation reduces CO2 and H+, increasing pH (respiratory alkalosis). Decreased ventilation increases CO2 and H+, decreasing pH (respiratory acidosis).
C. Renal Regulation of Acid-Base Balance
The kidneys fine-tune acid-base balance by excreting or reabsorbing H+ and HCO3-. They can also generate new HCO3- to compensate for acid overload.
D. Acid-Base Imbalances: Recognizing the Patterns
Acid-base imbalances are classified into four main categories:
-
Respiratory Acidosis: Elevated CO2 and decreased pH. Causes include hypoventilation (e.g., COPD, pneumonia, drug overdose).
-
Respiratory Alkalosis: Decreased CO2 and increased pH. Causes include hyperventilation (e.g., anxiety, high altitude).
-
Metabolic Acidosis: Decreased HCO3- and decreased pH. Causes include diabetic ketoacidosis, lactic acidosis, renal failure.
-
Metabolic Alkalosis: Increased HCO3- and increased pH. Causes include vomiting, diuretic use, and ingestion of excessive antacids.
IV. Clinical Implications and Nursing Management
Understanding fluid, electrolyte, and acid-base imbalances is crucial for accurate diagnosis and effective management. Nursing interventions focus on identifying the underlying cause, monitoring vital signs, assessing for symptoms, and administering appropriate treatments. This might include fluid replacement, electrolyte supplementation, medication administration (e.g., diuretics, bicarbonate), and respiratory support. Close monitoring of intake and output, electrolyte levels, and arterial blood gases (ABGs) are essential.
V. Preparing for ATI Quizzes and Exams
Success on ATI quizzes and exams requires a thorough understanding of the concepts discussed above. Here are some key strategies for effective learning and preparation:
-
Active Recall: Regularly test your knowledge using flashcards, practice questions, and self-testing.
-
Concept Mapping: Create visual representations of the interconnectedness of fluid, electrolyte, and acid-base balance.
-
Case Studies: Work through case studies to apply your knowledge to real-world scenarios.
-
Collaborative Learning: Study with peers to discuss challenging concepts and reinforce learning.
-
Review ATI Resources: Utilize any study guides, practice questions, and learning materials provided by ATI.
VI. Conclusion: Mastering the Essentials
Fluid, electrolyte, and acid-base balance are fundamental physiological processes. A deep understanding of these intricate mechanisms is essential for healthcare professionals. By mastering the core concepts and employing effective study strategies, you can confidently navigate challenging ATI quizzes and exams, and ultimately provide safe and effective patient care. Remember, consistent review and application of knowledge are key to success. Good luck!
Latest Posts
Latest Posts
-
Receptacles Listed As Tamper Resistant Are Not Required In
Apr 04, 2025
-
The Study Of How Wealth Is Created And Distributed Is
Apr 04, 2025
-
Es Mas Importante Doblar La Ropa Que Hablar Por Telefono
Apr 04, 2025
-
After Germanys Unification In 1871 Industrialization Accelerated And
Apr 04, 2025
-
Which Nursing Theory Focuses On The Clients Self Care Needs
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Fluid Electrolyte And Acid-base Balance Ati Quizlet . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
