Model 1 Movement Of Water In And Out Of Cells
Breaking News Today
Apr 04, 2025 · 6 min read
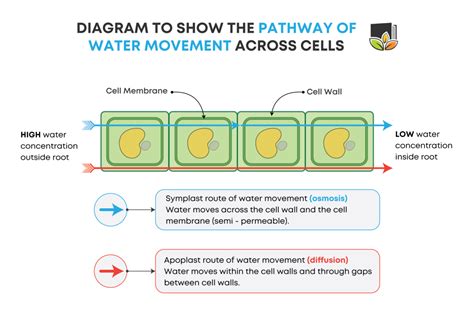
Table of Contents
Model 1: Movement of Water in and Out of Cells – Osmosis and Beyond
Understanding how water moves into and out of cells is fundamental to comprehending cellular function and overall organismal health. This article delves deep into Model 1, exploring the intricate processes governing water transport, focusing on osmosis, but also expanding to encompass other crucial mechanisms like aquaporins and the implications of water movement in different environments.
Osmosis: The Driving Force of Water Movement
At the heart of water transport in and out of cells lies osmosis, a special case of passive transport. Osmosis is the net movement of water molecules across a selectively permeable membrane from a region of high water concentration to a region of low water concentration. This movement continues until equilibrium is reached, meaning the water concentration is equal on both sides of the membrane. Crucially, this movement is driven by the water potential, a measure of the tendency of water to move from one area to another.
Water Potential: Understanding the Driving Force
Water potential is influenced by several factors:
-
Solute potential (Ψs): This represents the effect of dissolved solutes on water potential. The presence of solutes lowers water potential because solutes bind water molecules, reducing their free movement. A higher solute concentration results in a lower (more negative) solute potential.
-
Pressure potential (Ψp): This reflects the physical pressure on the water. Positive pressure potential (turgor pressure) occurs when water enters a cell, causing it to swell against its cell wall (in plant cells) or cell membrane (in animal cells). Negative pressure potential occurs when water leaves a cell, causing it to shrink.
-
Total water potential (Ψ): This is the sum of solute potential and pressure potential (Ψ = Ψs + Ψp). Water always moves from an area of higher water potential to an area of lower water potential.
Osmotic Environments and Their Effects on Cells
The environment surrounding a cell significantly impacts water movement. Three primary osmotic environments exist:
-
Isotonic solution: The solute concentration outside the cell is equal to the solute concentration inside the cell. Water moves in and out of the cell at equal rates, resulting in no net change in cell volume. This is often the ideal condition for animal cells.
-
Hypotonic solution: The solute concentration outside the cell is lower than the solute concentration inside the cell. Water moves into the cell, causing it to swell. In plant cells, this leads to turgor pressure, which helps maintain cell rigidity. Animal cells, however, can burst (lyse) under hypotonic conditions due to the lack of a rigid cell wall.
-
Hypertonic solution: The solute concentration outside the cell is higher than the solute concentration inside the cell. Water moves out of the cell, causing it to shrink (plasmolysis in plant cells, crenation in animal cells). This can severely disrupt cellular function and lead to cell death.
Beyond Osmosis: Aquaporins and Facilitated Water Transport
While osmosis explains the passive movement of water, the rate of water transport across cell membranes is often far higher than what simple diffusion across the lipid bilayer can account for. This is where aquaporins come into play.
Aquaporins: Specialized Water Channels
Aquaporins are integral membrane proteins that form channels selectively permeable to water molecules. They significantly increase the permeability of cell membranes to water, facilitating rapid water movement across the membrane. This is particularly important in cells that experience high rates of water transport, such as kidney cells and root hair cells.
Aquaporin Regulation and its Significance
The activity of aquaporins can be regulated, allowing cells to control the rate of water transport in response to changing conditions. This regulation can involve:
-
Phosphorylation: Adding a phosphate group can alter aquaporin conformation and influence its permeability.
-
Hormonal control: Hormones can trigger signaling pathways that modulate aquaporin activity.
-
Changes in pH: Fluctuations in pH can also affect aquaporin function.
This dynamic regulation of aquaporin activity is essential for maintaining cellular homeostasis and responding to osmotic stress.
Water Movement in Different Cell Types and Organisms
The mechanisms of water movement and their impact vary considerably depending on the type of cell and the organism.
Plant Cells: The Importance of Turgor Pressure
Plant cells possess a rigid cell wall that provides structural support. When water enters a plant cell via osmosis, it creates turgor pressure, pushing the cell membrane against the cell wall. This turgor pressure is crucial for maintaining cell shape, rigidity, and overall plant structure. Wilting occurs when water loss leads to a decrease in turgor pressure.
Animal Cells: Maintaining Osmotic Balance
Animal cells lack a rigid cell wall, making them highly susceptible to osmotic imbalances. Maintaining a stable internal osmotic environment is vital for their survival. This is achieved through various mechanisms, including the regulation of ion transport and the use of aquaporins. Disruptions in osmotic balance can lead to cell damage or death.
Specialized Cells and Tissues: Adapting to Water Stress
Certain cells and tissues are adapted to survive in extreme osmotic environments. For example, cells in the kidneys play a crucial role in regulating water balance in the body. They use aquaporins and other transport mechanisms to reabsorb water from the filtrate, preventing excessive water loss. Similarly, cells in desert plants have evolved mechanisms to minimize water loss in arid conditions.
Clinical and Environmental Relevance of Water Movement
Understanding the mechanisms of water transport has profound implications in various fields.
Medical Implications: Disorders Related to Water Imbalance
Several medical conditions stem from disruptions in water balance. For example:
-
Dehydration: Excessive water loss leads to cellular dehydration and impaired organ function.
-
Edema: Fluid accumulation in tissues due to impaired water regulation.
-
Hypernatremia and Hyponatremia: Disorders characterized by abnormally high or low sodium concentrations in the blood, disrupting osmotic balance.
-
Cystic fibrosis: A genetic disorder affecting ion transport, leading to imbalances in water regulation within the lungs and other organs.
Environmental Relevance: Plant responses to drought stress
Water availability is a critical factor influencing plant growth and survival. Drought stress leads to water loss from plant cells, causing wilting and potentially leading to plant death. Understanding how water moves in and out of plant cells under drought conditions is important for developing strategies to enhance crop resilience to drought.
Conclusion: A Dynamic and Crucial Process
The movement of water in and out of cells is a complex and dynamic process governed by osmosis, aquaporins, and other regulatory mechanisms. This process is fundamental to maintaining cellular homeostasis, supporting cellular function, and ensuring organismal survival. Disruptions in water movement can have severe consequences, highlighting the importance of understanding these intricate mechanisms. Further research continues to unravel the finer details of water transport and its implications for human health and environmental sustainability. The study of water movement in cells remains a vibrant field with significant implications for various scientific and clinical applications. Further research in this area promises to provide more detailed and refined models, allowing us to better understand and manage the intricacies of cellular hydration and overall organismal health. The continued exploration of this topic promises to enhance our understanding of basic cellular biology and help us develop more effective strategies for dealing with various health and environmental challenges.
Latest Posts
Latest Posts
-
Generally Speaking Electrical Cardioversion Is Not Recommended
Apr 05, 2025
-
Describe The Similarities And Differences Between Timelines And Flow Charts
Apr 05, 2025
-
Need For Refers To Maintaining Positive Relationships With Others
Apr 05, 2025
-
La Maison Es Un Mercado Al Aire Libre
Apr 05, 2025
-
Which Action Is One Role Of Rna Polymerase During Transcription
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Model 1 Movement Of Water In And Out Of Cells . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
