The Ich E6 Guideline Should Be Followed When:
Breaking News Today
Apr 05, 2025 · 6 min read
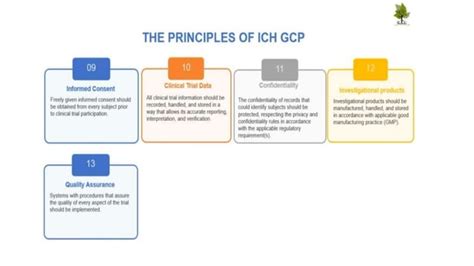
Table of Contents
The ICH E6 Guideline: When Should It Be Followed?
The ICH E6 guideline, formally titled "Good Clinical Practice (GCP) E6(R2)", provides a standardized framework for designing, conducting, recording, and reporting clinical trials involving human subjects. Its primary goal is to ensure the rights, safety, and well-being of trial participants while also generating credible and reliable data. But when exactly should you follow ICH E6? Understanding this crucial question is essential for anyone involved in clinical research. This article delves into the specific circumstances where adherence to ICH E6 is not only recommended but often mandated.
Understanding the Scope of ICH E6
Before exploring the "when," let's clarify the "what." ICH E6 isn't a mere suggestion; it's a comprehensive set of guidelines that establish ethical and scientific standards for clinical trials. These standards cover various aspects, including:
- Ethical Considerations: Protecting the rights and welfare of participants is paramount. This includes informed consent, data privacy, and the ability to withdraw from the trial at any time without penalty.
- Study Design and Conduct: ICH E6 outlines requirements for protocol development, investigator selection, site management, and data handling. It emphasizes the need for meticulous planning and execution to minimize bias and ensure data integrity.
- Data Management and Reporting: The guideline covers data collection, storage, analysis, and reporting, ensuring transparency and accuracy. It also addresses the importance of maintaining comprehensive documentation throughout the trial.
- Regulatory Compliance: Adherence to ICH E6 is often a prerequisite for regulatory approval of clinical trials in many countries. This means non-compliance can lead to delays, rejection of applications, and even legal repercussions.
Situations Requiring Strict Adherence to ICH E6
The ICH E6 guideline applies to a broad range of clinical trials, but its applicability isn't universal. Let's examine specific situations where strict adherence is absolutely crucial:
1. Trials Subject to Regulatory Oversight
This is perhaps the most straightforward scenario. Any clinical trial intending to submit data to regulatory authorities for drug approval or licensing must strictly adhere to ICH E6. This includes trials evaluating pharmaceuticals, biologics, medical devices, and other healthcare interventions. Regulatory agencies like the FDA (United States), EMA (European Union), and PMDA (Japan) explicitly require GCP compliance, making ICH E6 adherence non-negotiable. Failure to comply can lead to:
- Rejection of marketing applications: Data generated from non-compliant trials are deemed unreliable and will not be considered for approval.
- Audits and inspections: Regulatory bodies may conduct audits to verify GCP compliance. Non-compliance can result in warnings, fines, and even suspension of research activities.
- Legal ramifications: In some cases, serious breaches of GCP can lead to legal action.
2. Investigator-Initiated Trials (IITs)
Even trials initiated by individual researchers, often with smaller budgets and scope, must generally adhere to ICH E6. While the level of scrutiny might be slightly less intense compared to large, industry-sponsored trials, the fundamental principles of GCP still apply. The ethical considerations and data integrity standards remain paramount, irrespective of funding source. Non-compliance in IITs can still lead to:
- Publication rejection: Journals often require evidence of GCP compliance before accepting manuscripts for publication.
- Reputational damage: Non-compliance can severely damage the researcher's reputation and credibility within the scientific community.
- Funding difficulties: Future funding applications might be impacted by past non-compliance.
3. Multicenter Trials
Multicenter trials, involving multiple research sites and investigators, pose unique challenges to maintaining consistency and data integrity. ICH E6 is particularly critical in such settings to ensure standardized procedures and data management across all participating sites. Without rigorous adherence, the following issues can arise:
- Data inconsistencies: Different sites may employ different procedures, leading to inconsistencies and inaccuracies in the data.
- Difficulties in data analysis: Harmonizing data from various sites becomes significantly more complex when GCP principles are not consistently applied.
- Increased risk of bias: Inconsistencies in data collection and management can introduce bias into the study results.
4. Trials Involving Vulnerable Populations
Trials involving vulnerable populations, such as children, elderly individuals, pregnant women, or those with cognitive impairments, require an even higher level of ethical scrutiny. ICH E6 plays a crucial role in ensuring that the specific needs and vulnerabilities of these populations are adequately protected. Special considerations include:
- Informed consent procedures: Obtaining informed consent from vulnerable participants requires careful consideration and might involve legal guardians or representatives.
- Risk-benefit assessment: A meticulous assessment of potential risks and benefits is critical to ensure that the participation of vulnerable individuals is justified.
- Data privacy and confidentiality: Protecting the privacy and confidentiality of vulnerable participants is of paramount importance.
5. Trials Utilizing Novel Technologies or Methodologies
With the advent of new technologies and methodologies in clinical research (e.g., telehealth, wearable sensors, artificial intelligence), ensuring data integrity and patient safety becomes more complex. ICH E6 serves as a flexible framework that can be adapted to accommodate these advances while upholding the core principles of GCP. This adaptation requires careful consideration of:
- Data security and integrity: Novel technologies may present unique challenges related to data security and integrity, requiring robust measures to protect data confidentiality and prevent manipulation.
- Validation of new technologies: Appropriate validation and verification processes must be implemented to ensure the reliability and accuracy of data collected using new technologies.
- Ethical implications of new technologies: The ethical implications of using novel technologies in clinical research must be carefully considered and addressed.
Situations Where ICH E6 Might Not Be Strictly Applicable (With Cautions)
While ICH E6 is the gold standard for most clinical trials, there are a few exceptions or situations where its strict application might be relaxed, but with significant caveats and careful justification:
- Certain observational studies: Some observational studies, particularly those with minimal intervention, might not fall under the strict requirements of ICH E6. However, ethical considerations and data integrity must still be paramount. Transparency and rigorous methodology are key.
- Very early-phase research: In extremely early-phase research (e.g., pre-clinical studies involving only healthy volunteers, with minimal risk), some aspects of ICH E6 might be less applicable. However, ethical review and appropriate safety monitoring are still crucial.
- Qualitative research: Qualitative research, focusing on in-depth understanding of experiences and perspectives, might not require full adherence to all aspects of ICH E6. Nevertheless, principles of ethical conduct, informed consent, and data confidentiality remain essential.
It's crucial to note that even in these exceptions, deviations from ICH E6 should be clearly documented and justified. Ethical review boards (ERBs) or Institutional Review Boards (IRBs) play a vital role in reviewing and approving research protocols, even in these less strictly regulated contexts.
Conclusion: Prioritizing Ethical Conduct and Data Integrity
The ICH E6 guideline is more than just a set of rules; it's a reflection of a commitment to ethical research practices and the generation of trustworthy data. While there might be some nuanced situations where strict adherence to every aspect of the guideline might not be fully applicable, the overarching principles of protecting participant welfare, maintaining data integrity, and ensuring transparency should always remain the primary focus in any research endeavor involving human subjects. Understanding the specific contexts where ICH E6 applies is crucial for researchers, sponsors, and regulatory bodies alike to ensure ethical and scientifically robust clinical trials. Ignoring this guideline can have significant repercussions, jeopardizing the integrity of research and potentially harming participants. Therefore, prioritizing compliance with ICH E6 remains a fundamental requirement for responsible and successful clinical research.
Latest Posts
Latest Posts
-
In General Oral Glucose Should Be Given
Apr 05, 2025
-
Successful Leaders Allow Others To Take Risks
Apr 05, 2025
-
The Two Crucial Elements Of A Companys Business Model Are
Apr 05, 2025
-
According To The Nhtsa The Combination Of
Apr 05, 2025
-
Which Activities May Be Part Of A Campaign
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about The Ich E6 Guideline Should Be Followed When: . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
