Where Does Most Exogenous Antigen Presentation Take Place
Breaking News Today
Apr 02, 2025 · 6 min read
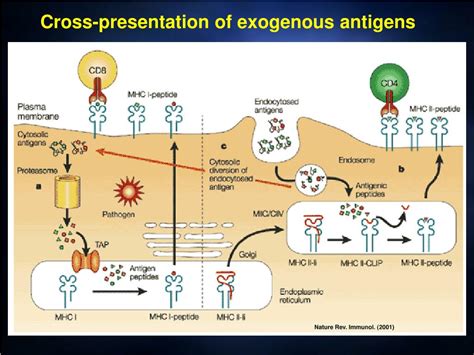
Table of Contents
Where Does Most Exogenous Antigen Presentation Take Place?
The immune system's ability to recognize and neutralize foreign invaders, or antigens, is paramount to maintaining health. A critical process in this defense mechanism is antigen presentation, the process by which specialized cells display fragments of antigens on their surface, alerting other immune cells to their presence. This presentation is broadly categorized into exogenous and endogenous pathways, depending on the origin of the antigen. This article focuses on exogenous antigen presentation, detailing where it predominantly occurs and the intricate cellular mechanisms involved.
Understanding Exogenous Antigen Presentation
Exogenous antigen presentation primarily involves the processing and presentation of antigens originating outside the cell. These antigens are typically extracellular pathogens, such as bacteria, viruses, fungi, and parasites, or even proteins from environmental sources. Unlike endogenous antigens, which originate within the cell, exogenous antigens require a distinct pathway for processing and presentation.
Key Players in Exogenous Antigen Presentation
The primary cell type responsible for exogenous antigen presentation is the dendritic cell (DC). DCs are professional antigen-presenting cells (APCs) with exceptional antigen-capture capabilities. Other APCs, including macrophages and B cells, also participate in exogenous antigen presentation, albeit to a lesser extent than DCs. These cells express major histocompatibility complex class II (MHC II) molecules on their surface, crucial for displaying processed antigen fragments to CD4+ T helper cells.
The Location of Exogenous Antigen Presentation: A Multi-Compartmental Process
The location of exogenous antigen presentation isn't confined to a single anatomical site. Instead, it's a dynamic and multi-stage process occurring in various locations throughout the body, each playing a crucial role in the initiation and amplification of the immune response.
1. The Site of Antigen Encounter: Peripheral Tissues
The initial encounter between exogenous antigens and APCs occurs primarily in peripheral tissues. These tissues act as the frontline defense, where antigens are first encountered. Examples include:
- Skin: Langerhans cells, a type of DC residing in the epidermis, are the first responders to skin infections. They capture antigens and migrate to draining lymph nodes for presentation.
- Mucosal Tissues (Gut, Lungs, etc.): Specialized DCs and macrophages reside in the mucosal linings of the gut, lungs, and other mucosal surfaces. They capture antigens from ingested food, inhaled air, and other sources.
- Connective Tissues: DCs and macrophages patrol the connective tissues, capturing antigens from invading pathogens or damaged cells.
At these peripheral sites, APCs efficiently capture antigens through various mechanisms:
- Phagocytosis: APCs engulf entire pathogens or particles.
- Pinocytosis: APCs take up fluids and dissolved materials.
- Receptor-mediated endocytosis: APCs bind to antigens via specific receptors and internalize them.
2. Migration to Lymph Nodes: The Central Hub of Antigen Presentation
Following antigen capture, APCs, particularly DCs, undergo a crucial maturation process. This involves upregulation of MHC II molecules and co-stimulatory molecules, which are essential for T cell activation. These mature APCs then migrate from the peripheral tissues to the draining lymph nodes.
Lymph nodes serve as central hubs for the adaptive immune response. They provide a structured environment where APCs efficiently present antigens to T cells. The high concentration of lymphocytes within the lymph nodes enhances the probability of successful antigen presentation and T cell activation. The microenvironment of the lymph node, including chemokines and cytokines, further facilitates this interaction.
3. Lymph Node Architecture: Specific Zones for Antigen Presentation
Within the lymph node, distinct microenvironments influence antigen presentation. Key areas include:
- T cell zones: Mature DCs migrate to the T cell zones, where they specifically interact with naive CD4+ T cells. The interaction between the MHC II-antigen complex on the DC and the T cell receptor (TCR) on the T cell, along with co-stimulatory signals, initiates T cell activation and differentiation.
- B cell zones: While primarily associated with B cell activation, the B cell zones also interact indirectly with exogenous antigen presentation. Helper T cells, activated in the T cell zones following antigen presentation by DCs, migrate to the B cell zones, assisting in B cell activation and antibody production.
4. Splenic Antigen Presentation: A Specialized Microenvironment
The spleen, in addition to lymph nodes, plays a significant role in exogenous antigen presentation, particularly for blood-borne antigens. DCs and macrophages within the spleen capture antigens from the blood and present them to T cells within the splenic white pulp, a region rich in lymphocytes. The architecture of the spleen facilitates efficient antigen trapping and presentation to circulating lymphocytes.
Detailed Mechanisms of Exogenous Antigen Processing
The efficiency of exogenous antigen presentation relies heavily on the processing of antigens into smaller peptides, which are then bound to MHC II molecules. This process involves several key steps:
-
Antigen Uptake: As previously discussed, APCs internalize antigens through various mechanisms.
-
Antigen Degradation: The internalized antigen is directed towards the endosomal/lysosomal pathway. Here, a series of hydrolytic enzymes, including proteases, break down the antigen into smaller peptides. The acidic environment of the endosome/lysosome is crucial for optimal antigen degradation.
-
MHC II Synthesis and Trafficking: MHC II molecules are synthesized in the endoplasmic reticulum (ER). They bind to an invariant chain, which prevents premature peptide binding.
-
Peptide Loading: The MHC II-invariant chain complex traffics to the endosome/lysosome, where the invariant chain is degraded, leaving a CLIP fragment bound to the MHC II molecule. A specialized molecule called HLA-DM exchanges CLIP with the processed antigen peptides.
-
MHC II-Peptide Complex Surface Expression: The stable MHC II-peptide complex is then transported to the cell surface for presentation to T cells.
Beyond DCs: The Role of Other APCs
While DCs are the most potent APCs for exogenous antigen presentation, other cells contribute significantly:
- Macrophages: Macrophages, abundant in various tissues, participate in antigen uptake and processing, presenting antigens to T cells. Their presentation, however, is generally less efficient than that of DCs.
- B cells: B cells, through their B cell receptor (BCR), can bind specific antigens and internalize them. They then process and present these antigens to T cells, crucial for B cell activation and antibody production. However, their antigen-presenting capacity is relatively specialized.
The Importance of Co-stimulation in Exogenous Antigen Presentation
Successful T cell activation requires not only MHC II-antigen presentation but also co-stimulation. Mature APCs express co-stimulatory molecules, such as B7 (CD80/CD86), which interact with CD28 on T cells. This co-stimulatory signal, alongside the TCR-MHC II interaction, is essential for full T cell activation. Without co-stimulation, T cells become anergic, unable to respond to subsequent antigen encounters. The importance of co-stimulation highlights the critical role of APC maturation in effective immune responses.
Clinical Implications and Future Directions
Understanding the intricacies of exogenous antigen presentation is crucial for developing effective vaccines and immunotherapies. Targeting specific aspects of antigen processing and presentation pathways could enhance the efficacy of these strategies. Future research will likely focus on:
- Developing more effective vaccines: By optimizing antigen delivery and presentation, vaccines can elicit stronger and more durable immune responses.
- Improving cancer immunotherapy: Harnessing the power of DCs and other APCs to present tumor antigens could enhance anti-tumor immunity.
- Treating autoimmune diseases: Modulating the activity of APCs could help to suppress excessive immune responses in autoimmune diseases.
In conclusion, exogenous antigen presentation is a complex, multi-stage process primarily occurring in peripheral tissues and culminating in lymph nodes and the spleen. Dendritic cells are the major players, but macrophages and B cells also contribute. The efficient processing and presentation of antigens, along with crucial co-stimulatory signals, are essential for initiating and amplifying adaptive immune responses. Understanding these intricate mechanisms has profound implications for the development of advanced immunotherapies and vaccines.
Latest Posts
Latest Posts
-
Which Is An Appropriate Expected Outcome For A Client
Apr 03, 2025
-
A Complete And Accurate Medical Record Provides Legal Protection For
Apr 03, 2025
-
How Is Carbon Reintroduced Into The Atmosphere
Apr 03, 2025
-
Limestone And Marble Weather Faster Than Granite Because
Apr 03, 2025
-
Which Relationships Could Have A Negative Correlation Select Three Options
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Where Does Most Exogenous Antigen Presentation Take Place . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
