Why Is Water Considered To Be Polar
Breaking News Today
Apr 03, 2025 · 6 min read
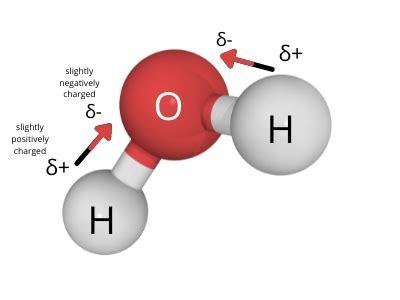
Table of Contents
Why is Water Considered to be Polar? Understanding Polarity and its Implications
Water, the elixir of life, is far more than just a simple compound of hydrogen and oxygen (H₂O). Its unique properties, crucial for supporting life as we know it, stem from a fundamental characteristic: polarity. Understanding why water is polar is key to grasping its extraordinary behavior and its vital role in various biological and chemical processes. This article delves deep into the concept of polarity, focusing specifically on the reasons behind water's polar nature and its far-reaching consequences.
Understanding Polarity: A Dip into Chemistry
Before we delve into water's polarity, let's establish a clear understanding of what "polar" means in a chemical context. Polarity arises from differences in electronegativity between atoms within a molecule. Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. When atoms with significantly different electronegativities bond, the more electronegative atom pulls the shared electrons closer, creating a partial negative charge (δ-) around it. Conversely, the less electronegative atom develops a partial positive charge (δ+). This uneven distribution of charge creates a dipole moment, essentially giving the molecule a positive and a negative end, like a tiny magnet.
The Electronegativity Difference in Water: Oxygen's Powerful Pull
The key to understanding water's polarity lies in the difference in electronegativity between oxygen and hydrogen. Oxygen is significantly more electronegative than hydrogen. This means that in each O-H bond of the water molecule, the oxygen atom attracts the shared electrons much more strongly than the hydrogen atoms. This unequal sharing of electrons results in a partial negative charge (δ-) on the oxygen atom and partial positive charges (δ+) on the two hydrogen atoms.
Visualizing the Water Molecule's Polarity
Imagine the water molecule as a bent structure, with the oxygen atom at the center and the two hydrogen atoms slightly offset. This bent geometry, along with the unequal distribution of charge, is crucial. If the molecule were linear, the dipole moments of the two O-H bonds would cancel each other out, resulting in a nonpolar molecule. However, the bent shape ensures that the dipole moments add up, creating a net dipole moment and making the water molecule distinctly polar.
Consequences of Water's Polarity: A World of Unique Properties
Water's polarity is responsible for many of its unusual and vital properties, influencing its behavior in numerous ways:
1. High Surface Tension: Water's Cohesive Strength
The strong intermolecular forces between water molecules, called hydrogen bonds, are a direct consequence of its polarity. The partially positive hydrogen atoms of one water molecule are attracted to the partially negative oxygen atoms of neighboring molecules. These hydrogen bonds create a strong cohesive force, holding the water molecules tightly together. This high cohesion leads to the high surface tension of water, enabling phenomena like water striders walking on water's surface.
2. High Boiling Point: Breaking the Hydrogen Bonds
The hydrogen bonds between water molecules also account for its relatively high boiling point compared to other molecules of similar size. A significant amount of energy is required to overcome these strong intermolecular attractions and break the hydrogen bonds, allowing the water molecules to transition from liquid to gas. This high boiling point is essential for maintaining liquid water on Earth's surface and supporting aquatic life.
3. Excellent Solvent: The Universal Solvent
Water's polarity makes it an excellent solvent for many ionic and polar substances. The partial charges on the water molecule can interact with and surround ions or polar molecules, effectively dissolving them. This dissolving power is crucial in biological systems, allowing for the transport of nutrients, the removal of waste products, and many other essential processes. This is why water is often referred to as the "universal solvent," although it's not truly universal as it doesn't dissolve everything.
4. High Specific Heat Capacity: Temperature Regulation
Water has a remarkably high specific heat capacity, meaning it can absorb a large amount of heat energy without experiencing a significant temperature change. This property is critical for regulating temperature, both in the environment and within living organisms. Large bodies of water, like oceans, act as massive heat reservoirs, moderating temperature fluctuations and creating more stable climates. Similarly, water within organisms helps to maintain stable internal temperatures.
5. Density Anomaly: Ice Floats
Water exhibits a unique density anomaly. Ice, the solid form of water, is less dense than liquid water. This is because the hydrogen bonds in ice form a more open, crystalline structure compared to liquid water. This lower density of ice allows it to float on liquid water, preventing lakes and oceans from freezing solid from the bottom up, preserving aquatic life.
6. Capillary Action: Water's Ascent
The combined effects of cohesion (water molecules sticking to each other) and adhesion (water molecules sticking to other surfaces) result in capillary action. Water molecules can move against gravity in narrow tubes or spaces, facilitated by both the hydrogen bonds and the attraction between water and the surface. This phenomenon is vital for transporting water in plants, through the xylem vessels, from roots to leaves.
Beyond the Basics: Exploring Deeper Implications
Water's polarity has ramifications far beyond these fundamental properties. Its influence extends to:
- Chemical reactions: Water's polarity plays a crucial role in countless chemical reactions, acting as a reactant, a solvent, or a medium for interactions. Many biochemical reactions within living organisms rely on water's polar nature.
- Biological membranes: The structure and function of cell membranes are heavily influenced by water's polarity. The hydrophobic (water-fearing) tails of phospholipids cluster together, while the hydrophilic (water-loving) heads interact with the surrounding water, creating a selectively permeable barrier.
- Protein folding: The three-dimensional structure of proteins, crucial for their function, is significantly impacted by interactions between the polar and nonpolar amino acid side chains and water molecules.
- Climate regulation: The polar nature of water influences global climate patterns through its roles in evaporation, condensation, precipitation, and heat transfer.
Misconceptions and Clarifications
It's important to address some common misconceptions related to water's polarity:
- Water is not fully ionized: While water molecules can undergo self-ionization to a small extent (producing H⁺ and OH⁻ ions), the majority of water molecules remain intact. The partial charges are responsible for the polarity, not the complete dissociation into ions.
- Polarity is not just about hydrogen bonds: While hydrogen bonds are a significant consequence of water's polarity, polarity itself is a fundamental property arising from the electronegativity difference between oxygen and hydrogen.
Conclusion: The Polarity of Life
In conclusion, water's polarity is not merely a chemical detail; it's the foundation of many of its exceptional properties. This seemingly simple molecule, with its unique polar character, is the cornerstone of life on Earth, driving numerous biological and chemical processes. Understanding the intricacies of water's polarity provides invaluable insight into the fundamental workings of the natural world and the incredible role this essential substance plays in maintaining life as we know it. Further research continues to reveal more intricate details about the impact of water's polarity, constantly expanding our comprehension of its significance in the universe.
Latest Posts
Related Post
Thank you for visiting our website which covers about Why Is Water Considered To Be Polar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
