First 30 Elements On The Periodic Table
Breaking News Today
Apr 02, 2025 · 6 min read
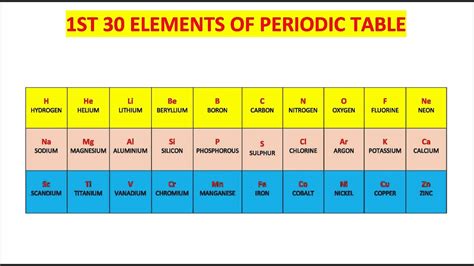
Table of Contents
The First 30 Elements: A Deep Dive into the Building Blocks of Matter
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic number and recurring chemical properties. This article delves into the fascinating world of the first 30 elements, exploring their properties, uses, and significance in our daily lives and the wider universe. Understanding these fundamental building blocks is crucial to grasping the complexity of the world around us. We'll examine each element individually, highlighting key characteristics and practical applications.
The First 10 Elements: The Foundation of Chemistry
The first ten elements form the bedrock of the periodic table, representing the simplest atomic structures. Their properties largely define the behavior of more complex elements and molecules.
Hydrogen (H): Atomic Number 1
- Properties: Colorless, odorless, tasteless, highly flammable diatomic gas. The most abundant element in the universe.
- Uses: Ammonia production (fertilizers), petroleum refining, fuel for rockets, and emerging applications in fuel cells. Isotopes like deuterium and tritium have applications in nuclear research and energy.
Helium (He): Atomic Number 2
- Properties: Inert, colorless, odorless, tasteless noble gas. The second most abundant element in the universe. Very low density.
- Uses: Balloons, cryogenics (cooling superconducting magnets in MRI machines), leak detection, and welding.
Lithium (Li): Atomic Number 3
- Properties: Soft, silvery-white alkali metal. Highly reactive.
- Uses: Batteries (lithium-ion batteries power many electronic devices), ceramics, glass, and lubricating greases. Also used in some psychiatric medications.
Beryllium (Be): Atomic Number 4
- Properties: Steel-gray, lightweight, alkaline earth metal. Toxic and carcinogenic. High strength-to-weight ratio.
- Uses: Aerospace applications (lightweight components), nuclear reactors (neutron reflectors), and specialized tools.
Boron (B): Atomic Number 5
- Properties: Metalloid, hard, brittle, and a poor conductor of electricity.
- Uses: Fiberglass (reinforcement in composites), insecticides, and detergents. Boron compounds are used in nuclear applications and in semiconductors.
Carbon (C): Atomic Number 6
- Properties: Nonmetal, tetravalent, existing in various allotropes (diamond, graphite, fullerene). Basis of all organic life.
- Uses: Fuels, plastics, building materials (diamonds), pencils (graphite), and countless other applications due to its versatile bonding capabilities.
Nitrogen (N): Atomic Number 7
- Properties: Colorless, odorless, tasteless diatomic gas. Makes up about 78% of Earth's atmosphere. Relatively inert.
- Uses: Fertilizers (ammonia production), food preservation, and the production of various chemicals.
Oxygen (O): Atomic Number 8
- Properties: Colorless, odorless, tasteless diatomic gas. Essential for respiration in most living organisms. Highly reactive.
- Uses: Respiration, combustion, and various industrial processes (metal production, chemical synthesis).
Fluorine (F): Atomic Number 9
- Properties: Pale yellow, highly reactive diatomic gas. The most electronegative element.
- Uses: Production of fluorocarbons (refrigerants, plastics), toothpaste (fluoride), and in etching glass.
Neon (Ne): Atomic Number 10
- Properties: Inert, colorless, odorless, tasteless noble gas. Glows brightly when an electric current is passed through it.
- Uses: Neon signs, lasers, and cryogenics.
Elements 11-20: Expanding the Chemical Landscape
This section explores elements that showcase a broader range of properties and applications, from the essential sodium to the reactive phosphorus.
Sodium (Na): Atomic Number 11
- Properties: Soft, silvery-white alkali metal. Highly reactive with water.
- Uses: Table salt (sodium chloride), street lighting (sodium-vapor lamps), and in many industrial chemicals.
Magnesium (Mg): Atomic Number 12
- Properties: Lightweight, silvery-white alkaline earth metal. Burns with a bright white light.
- Uses: Alloying agent in aluminum and other metals, making lightweight structural components, and in flares and fireworks.
Aluminum (Al): Atomic Number 13
- Properties: Lightweight, ductile, and relatively inexpensive metal. Good conductor of electricity.
- Uses: Packaging (cans), building materials, transportation (vehicles), and electrical wiring.
Silicon (Si): Atomic Number 14
- Properties: Metalloid, hard, brittle, and a semiconductor. The second most abundant element in the Earth's crust.
- Uses: Semiconductors (computer chips, solar cells), glass, ceramics, and silicones.
Phosphorus (P): Atomic Number 15
- Properties: Nonmetal, highly reactive, existing in several allotropes (white phosphorus is highly toxic and reactive).
- Uses: Fertilizers, detergents, and matches. Essential nutrient for living organisms.
Sulfur (S): Atomic Number 16
- Properties: Yellow, brittle nonmetal. Found in various allotropes.
- Uses: Sulfuric acid production, vulcanization of rubber, and in fertilizers and fungicides.
Chlorine (Cl): Atomic Number 17
- Properties: Greenish-yellow, highly reactive diatomic gas. A powerful disinfectant.
- Uses: Water purification (bleaching and disinfection), production of plastics (PVC), and in various industrial chemicals.
Argon (Ar): Atomic Number 18
- Properties: Inert, colorless, odorless, tasteless noble gas. Relatively abundant in the atmosphere.
- Uses: Protective atmosphere for welding and other high-temperature processes, and in fluorescent lighting.
Potassium (K): Atomic Number 19
- Properties: Soft, silvery-white alkali metal. Highly reactive with water.
- Uses: Fertilizers, potassium salts (various industrial applications), and in the human body (essential electrolyte).
Calcium (Ca): Atomic Number 20
- Properties: Soft, silvery-white alkaline earth metal. Relatively reactive.
- Uses: Cement, plaster, and in the human body (bones and teeth).
Elements 21-30: Transition Metals and Beyond
The final ten elements in this exploration showcase the increasing complexity of atomic structure and a greater diversity of properties and applications.
Scandium (Sc): Atomic Number 21
- Properties: Silvery-white transition metal. Relatively rare.
- Uses: High-intensity lighting, alloys, and catalysts.
Titanium (Ti): Atomic Number 22
- Properties: Strong, lightweight transition metal. Excellent corrosion resistance.
- Uses: Aerospace applications, medical implants, and sports equipment.
Vanadium (V): Atomic Number 23
- Properties: Hard, silvery-gray transition metal. Used in various alloys.
- Uses: Steel alloys (high-strength steels), and in some catalysts.
Chromium (Cr): Atomic Number 24
- Properties: Hard, silvery-white transition metal. Highly resistant to corrosion.
- Uses: Chrome plating, stainless steel, and pigments.
Manganese (Mn): Atomic Number 25
- Properties: Hard, brittle transition metal. Essential nutrient for humans and animals.
- Uses: Steel alloys (increases strength and hardness), and in fertilizers.
Iron (Fe): Atomic Number 26
- Properties: Silvery-gray, ductile metal. The most abundant metal in the Earth's crust. Ferromagnetic.
- Uses: Steel production, construction, and countless other applications. Essential for human life.
Cobalt (Co): Atomic Number 27
- Properties: Hard, brittle transition metal. Ferromagnetic.
- Uses: Magnetic alloys (magnets), catalysts, and in some medical applications.
Nickel (Ni): Atomic Number 28
- Properties: Silvery-white, ductile metal. Ferromagnetic. Corrosion resistant.
- Uses: Stainless steel, nickel plating, and catalysts.
Copper (Cu): Atomic Number 29
- Properties: Reddish-brown, ductile metal. Excellent conductor of electricity and heat.
- Uses: Electrical wiring, plumbing, and various alloys (brass, bronze).
Zinc (Zn): Atomic Number 30
- Properties: Bluish-white, brittle metal at room temperature, but ductile at higher temperatures. Corrosion resistant.
- Uses: Galvanizing steel (corrosion protection), brass production, and in various alloys. Essential nutrient for humans and animals.
Conclusion: The Significance of the First 30 Elements
The first 30 elements represent a crucial foundation in understanding chemistry and the materials that shape our world. From the simplest gases to the robust transition metals, each element plays a unique and vital role in various applications, from everyday life to high-tech industries. Understanding their properties and interactions is fundamental to advancements in science, technology, and medicine. Further exploration into the periodic table will reveal even more fascinating elements and their contributions to the universe. This deep dive into the first thirty elements serves as a gateway to a richer understanding of the fundamental building blocks of matter and their profound impact on our lives.
Latest Posts
Latest Posts
-
Allocating Common Fixed Expenses To Business Segments
Apr 03, 2025
-
What Percentage Of Your Gross Salary Does The Consumer Financial
Apr 03, 2025
-
Which Reason Best Explains Why Metals Are Shiny
Apr 03, 2025
-
Which Statement Best Describes What A Thylakoid Does During Photosynthesis
Apr 03, 2025
-
Unit 6 Consequences Of Industrialization Study Guide
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about First 30 Elements On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
